Alloys are metallic mixtures of two or more elements. For alloys to form spontaneously, there has to be an energy benefit to the elements mixing. We describe this energy benefit as the Gibbs free energy of mixing (Gmix). The nature of how Gmix changes with pressure, temperature, and the composition of the alloy can have a profound effect on how elements distribute themselves between different phases in nature. I am interested in how changes to Gmix in alloys can affect the distribution of trace elements between the core and mantle of Earth.
Alloys are a type of solution and, like other solutions, can be described as either ideal or non-ideal. Ideal solutions are the most simple. Let’s imagine an ideal solution of two components; A and B. In this solution, interactions between A-A, B-B, and A-B are all the same. This leads to some important features of ideal solutions. For example, in ideal solutions the volume change of the solution when A and B mix is a linear combination of the volumes of the pure phases of A and B (red line in Fig 1).
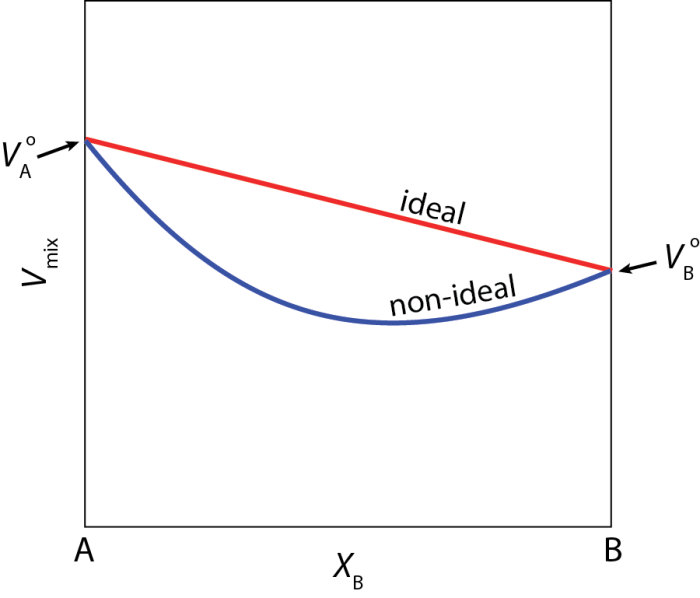
In non-ideal solutions, the interactions between A and B are not all the same. This leads to more complex changes in the solution properties upon mixing. Again looking at volume, we can see that for non-ideal solutions the volume change when A and B are mixed is not linear (blue line in Fig 1). Many alloys display non-ideal behavior like this.
It is common to describe the properties of non-ideal solutions relative to those of the equivalent ideal solution. These are termed the excess mixing properties of the solution. The excess Gibbs free energy of mixing is therefore defined as Gmix (excess) = Gmix (non-ideal) – Gmix (ideal). There are a variety of ways to describe Gmix (excess) as a function of pressure, temperature, and composition. In Figure 2, Gmix (excess) in shown for a solution of Fe and Pd as a function of composition and temperature using interaction parameters from Borisov & Palme (2000).

The same interaction parameters that describe Gmix (excess) can also be used to describe how the activity of each component in the alloy will vary. Activity can be thought of as the availability of a component for reaction or, as Wikipedia describes, an “effective concentration”. The activity and mole fraction of a component are related via the activity coefficient. Activity coefficients are important to describe how elements will partition between phases as a function of composition.
I have taken several different approaches to determine activity coefficients for alloy solutions. One method I have employed is to equilibrate alloys with a silicate melt under an atmosphere of controlled composition. Using this arrangement, I was able to determine the activity of Fe in Ir-Pt-Fe ternary alloys (Figure 3).

Clear from Figure 3 is that the real data do not behave as expected for an ideal solution. Instead, highly non-ideal behavior is observed. At low concentrations of Fe, the data appears to follow Henry’s law behavior – a linear relationship between concentration (mole fraction) and activity.
At higher pressures, we have to take a different approach. One way that we can probe the effect of alloy composition on trace element activity coefficients is to leverage the Soret effect. With this method, an alloy sample is compressed to high pressure and then heated so that it experiences a temperature gradient. The thermal gradient causes the major elements in the alloy to be redistributed, resulting in a chemical profile. The trace elements in the system are also redistributed, but not directly due to the thermal gradient. Instead, they respond to changes in the major element composition of the alloy along it’s length. At adjacent points along the sample, the trace element activity has to be approximately equal. If the activity of a trace element depends upon composition, then a concentration profile must develop in response to the major element gradient. Figure 4 demonstrates how this type of experiment is set up and the relationship between element concentration, activity, and activity coefficient:

In Figure 4, the major elements in the alloy are Fe, Ni, and O. The trace elements are the platinum group elements, Re, Au, W, and Mn. If you’re interested in a rigorous description of how these experiments work, how the activity coefficients are extracted, and the limitations of the approach, there are links at the bottom of the page to papers by Brenan & Bennett (2010) and Brenan, Bennett, and Zajacz (2016) that describe the approach in detail.
Associated Papers
Brenan, J. M., & Bennett, N. R. 2010. Soret separation of highly siderophile elements in Fe-Ni-S melts: Implications for solid metal-liquid metal partitioning, Earth and Planetary Science Letters, 298, 299-305.
Brenan, J. M., Bennett, N. R., Zacjacz, Z. 2016. Experimental Results on Fractionation of the Highly Siderophile Elements (HSE) at Variable Pressures and Temperatures during Planetary and Magmatic Differentiation, Reviews in Mineralogy and Geochemistry, 81.